What Is Actually Sitting on Your Scalp
- Hairline Illusions

- Mar 6
- 11 min read
Updated: Mar 17
The Science of Plastic Cap Materials, Dermal Absorption, and What Every Wig Wearer Needs to Know
You May Not Connect What You Are Feeling to What Is Sitting on Your Scalp

Many of the symptoms associated with long-term plastic chemical exposure are easy to dismiss or attribute to something else entirely. Unexplained fatigue. Skin irritation, redness, or scalp inflammation that comes and goes. Brain fog. Irregular menstrual cycles or hormonal shifts that appear without a clear cause. Difficulty with weight regulation. Thyroid irregularities. Reduced fertility. These are not rare or dramatic symptoms. They are the kind of things people mention at routine checkups and are often told are stress-related, age-related, or simply unexplained.
The chemicals found in plastic wig cap bases, including flame retardants, phthalates, and bisphenols, are classified as endocrine-disrupting chemicals. They do not cause immediate or obvious reactions. They accumulate. They interfere with the body's hormone signaling over time, affecting the thyroid, reproductive system, metabolism, immune function, and neurological development. The Endocrine Society, in a comprehensive report produced with international toxicology researchers, identified more than 1,000 manufactured chemicals currently in use as likely endocrine disruptors, with plastics identified as a primary and pervasive exposure source.
The scalp is not a barrier. For someone wearing a plastic-based wig or cranial prosthetic daily, often for 12 to 16 hours, often while warm and perspiring, chemical exposure is not theoretical. It is ongoing, cumulative, and happening against one of the body's most absorptive surfaces. What you are feeling may have more to do with what is sitting on your scalp than anyone has ever told you.
We have spent years educating professionals and clients about cap construction, scalp assessment, and material selection. But there is a conversation happening in the broader scientific community that the hair replacement industry has been largely absent from, and it is one we can no longer afford to ignore.
Plastics are in the human body. Not hypothetically. Not in trace amounts too small to matter. Microplastics have now been detected in human blood, lung tissue, placental tissue, and the lymphatic system. The scientific literature documenting this is growing rapidly, and what is emerging paints a clear picture: prolonged, direct skin contact with plastic-based materials is a meaningful exposure pathway.
When we place a polyurethane wig base, a plastic-coated monofilament cap, or a PU perimeter strip against a human scalp for hours at a time, day after day, we are not doing something neutral. We are making a clinical decision, whether we frame it that way or not.
This article examines the three most common plastic-containing cap constructions used in cranial prosthetics and hair replacement, the scientific evidence on what those materials can do in contact with human skin, and what professionals and clients can do to protect themselves.

The Three Cap Materials You Need to Understand
Lace
Lace caps, often described as the gold standard for natural appearance, are constructed from woven nylon or polyester mesh. The material itself is synthetic, and while lace is far more porous than solid plastic bases, the concern is not always the lace fiber in isolation.
The problem arises in how lace is used. Lace requires adhesives and tapes to be secured to the scalp. Many of these bonding agents contain acrylates, formaldehyde-releasing preservatives, and chemical solvents that sit directly on the skin. A 2025 study published in Environment & Health by researchers at the Silent Spring Institute analyzed 43 hair extension and synthetic hair products and detected 933 unique chemical signatures across the samples, identifying 169 distinct chemicals linked to cancer, hormone disruption, immune system effects, and developmental harm. Researchers noted that for millions of women, prolonged wear brings these substances into direct, continuous contact with the scalp and neck.
On an intact scalp, this is a concern worth tracking. On a compromised scalp, it is a clinical risk.
Monofilament
Monofilament is a thin, sheer fabric, often made from nylon or polyester, through which hair is individually hand-tied to create the appearance of natural scalp. It is widely regarded as one of the most comfortable cap materials due to its soft, skin-like finish.
The concern with monofilament is what it is coated or treated with during manufacturing. Many monofilament caps are backed or reinforced with polyurethane layers or bonded to other synthetic substrates to provide structure. The monofilament fabric itself, while thin and breathable, becomes a delivery system for whatever chemical treatments have been applied to it in the manufacturing process.
Additionally, monofilament traps heat and perspiration against the scalp. When skin is warm and moist, its permeability increases. Research published in Environment International in 2024 by the University of Birmingham demonstrated that sweatier, more hydrated skin absorbs significantly higher levels of chemical additives from plastic contact than dry skin. This is not a minor variable. It is a physiological reality that transforms a standard wear session into a higher-exposure event.
Polyurethane (PU)
Polyurethane is the most clinically significant cap material from a chemical exposure standpoint. PU bases are solid, non-porous plastic films derived from isocyanate and polyol chemistry. They are used as full base caps, perimeter strips, and bonded edges on hybrid cap constructions.
Polyurethane is valued in wig construction for its ability to hold hair knots securely, create a realistic scalp appearance, and provide a smooth adhesion surface. But it is also completely occlusive. A PU base does not breathe. It traps heat, moisture, and scalp secretions against the skin for the entire duration of wear.
The chemical composition of polyurethane includes residual isocyanates, plasticizers, and, in lower-grade formulations, phthalates and organotins. Phthalates are well-documented endocrine-disrupting chemicals. Organotins, detected in nearly 10% of synthetic hair products tested by Silent Spring Institute researchers, exceeded European Union safety limits in several samples and are associated with hormone disruption, reproductive toxicity, and immune system interference.
When PU is in continuous contact with warm, perspiring skin, these additives can leach into the skin surface film and, from there, into the body.
What the Science Says About Plastic and Human Skin
The dermal absorption of plastic additives is no longer theoretical. It is documented.
A landmark 2024 study published in Environment International, conducted by researchers at the University of Birmingham and Brunel University London, provided the first experimental evidence that chemical additives in microplastics can leach into human sweat and then be absorbed through the skin barrier into the bloodstream. Using 3D human skin equivalent models, researchers demonstrated that flame retardant chemicals (polybrominated diphenyl ethers, or PBDEs) present in polyethylene and polypropylene microplastics were bioavailable to the skin at rates of up to 8% of the exposure dose, with five specific compounds crossing the skin barrier to reach the equivalent of the bloodstream.
A related study published in Environmental Science & Technology established that skin hydration was a major driver of absorption, with sebum and sweat increasing the bioaccessibility of these chemicals from plastic surfaces. The scalp, which produces more sebum per unit area than most body surfaces and is regularly occluded by cap materials, represents an ideal absorption environment.
Research published in a 2024 review in MDPI's Cosmetics journal on microplastic and nanoplastic penetration through skin confirmed that smaller plastic particles, particularly those in the nanoscale range below 100 nanometers, can penetrate through the dermal barrier and accumulate in epidermal Langerhans cells, particularly around hair follicles. The follicular pathway, the same pathway through which many topical medications are designed to be delivered, is also a pathway for chemical absorption from materials in sustained contact with the scalp.
A 2024 study in Environment International also found that aged polystyrene microplastics exacerbate alopecia through tight junction injuries and apoptosis via oxidative stress pathways in skin. The connection between plastic exposure and hair loss mechanisms is emerging as an active area of research.
What all of this tells us is that the scalp is not a passive surface. It is an active organ with absorptive capacity, and what we place against it for hours at a time contributes to the body's total chemical load.
The Broader Context: Plastics in the Body
The hair replacement industry does not exist in isolation from the broader scientific conversation about human plastic exposure. That context matters.
Microplastics have now been detected in human blood, lung tissue, testicular tissue, placental tissue, and breast milk. A 2022 study in Environment International by researchers at Vrije Universiteit Amsterdam confirmed microplastics in the blood of 17 of 22 anonymous healthy donors. The most common polymer types found were polyethylene terephthalate (PET), polystyrene, and polyethylene, all of which have applications in textile and synthetic hair manufacturing.
The National Institute of Environmental Health Sciences has identified endocrine-disrupting chemicals, including those found in plastic formulations, as capable of negatively affecting immunity, metabolism, puberty timing, and reproductive function even at low levels. These effects are cumulative and do not require a single high-dose exposure event to manifest.
A 2025 Consumer Reports study found dangerous chemicals in 100% of braiding hair samples tested, including benzene, a known carcinogen, in 30% of products, and lead in 90% of samples. Researcher Tamarra James-Todd of Harvard T.H. Chan School of Public Health noted that these chemicals do not operate in isolation. They sit on the scalp, where they can be dermally absorbed, inhaled, and transferred via hand-to-mouth contact.
The cumulative load matters. A client who wears a PU-based cranial prosthetic daily, uses adhesive to bond it, applies edge control with synthetic polymers, and has done so for months or years is not being exposed to a single source. They are accumulating exposure across multiple vectors simultaneously, with the scalp as the primary contact surface.
Compromised Scalps Carry Greater Risk
For general wig wearers on healthy scalps, the chemical exposure from cap materials is a background concern worth monitoring. For medically vulnerable clients, it rises to a clinical priority.
Chemotherapy disrupts the skin barrier. Radiation damages the microvasculature and impairs the skin's ability to regulate absorption. Autoimmune conditions involve immune dysregulation that can amplify inflammatory responses to chemical exposures. Post-surgical scalps have compromised tissue integrity at the incision and treatment areas.
The University of Birmingham's 2024 findings established that sweatier, more hydrated skin absorbs significantly higher concentrations of plastic additives than dry skin. A client undergoing treatment, whose scalp is already compromised, warm, and potentially moist from perspiration or topical medications, is wearing a cap under conditions that maximize absorption.
This is the clinical reality that separates cranial prosthetics from cosmetic wig service. The professional who does not understand this distinction cannot adequately protect their clients.
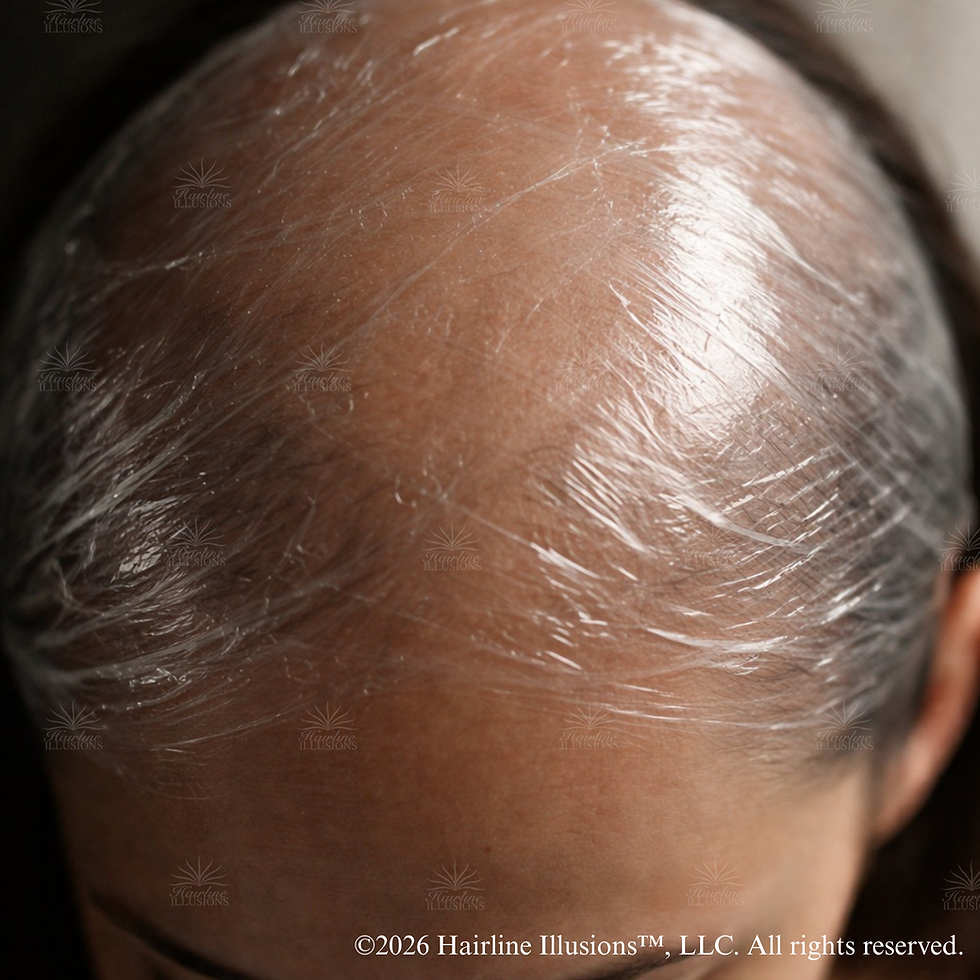
A Note on Plastic Molds During the Fitting Process
A reasonable question arises in clinical practice: if prolonged plastic contact carries chemical absorption risk, what about the short-term plastic used during impression or mold-making sessions?
Duration matters, but it does not eliminate risk entirely. Sweat is the mechanism that activates chemical leaching from plastic materials. The sebum in sweat facilitates the release of toxic chemicals out of plastic and renders them available for absorption through the skin. A mold session typically lasts minutes, not hours, which reduces cumulative exposure. However, clients who are post-chemotherapy, receiving radiation, or managing autoimmune conditions often present with compromised scalp integrity, meaning the standard assumptions about short-term exposure may not apply to them. The scalp is also among the highest-absorbing regions of the body, alongside the forehead and armpit.
For a client with an intact, healthy scalp in a climate-controlled environment, short-term mold contact carries low risk. For a client who is warm, sweating, or has a compromised scalp, the prudent standard is to use ISO 10993 biocompatible-rated impression materials, minimize contact time, and avoid leaving any plastic material against the scalp outside the active impression window. Short-term does not mean zero risk. It means the risk is manageable when we make informed material choices.
What Professionals and Clients Can Do
Prioritize Biocompatible and Certified Materials
When selecting cap constructions for medically vulnerable clients, material choice must be evaluated against biocompatibility standards. ISO 10993, the international standard for biological evaluation of medical devices, establishes a framework for assessing whether materials are safe for prolonged skin contact. Cap materials that have not been evaluated within this framework should not be the default choice for clients with compromised scalps.
Look for monofilament and lace constructions from manufacturers who can document material sourcing and disclose chemical treatments. Avoid cap bases that combine PU with monofilament or lace without full material disclosure. For clients who require PU components, limit the surface area of PU contact and ensure it does not cover the entire scalp.
Use a Cap Liner. Choose It Carefully.
A cap liner, worn between the scalp and the wig, is one of the most effective and underutilized tools for reducing direct chemical contact. However, the liner itself must be selected with the same scrutiny we apply to the wig.
The most appropriate cap liner materials for sensitive or medically compromised scalps are natural and minimally processed. Bamboo is highly regarded for its softness, moisture-wicking properties, and natural antimicrobial characteristics. Organic cotton is another excellent option, providing breathability, gentleness on fragile skin, and a physical barrier between the scalp and synthetic cap materials. Silk is appropriate for clients with significant scalp sensitivity or post-radiation skin due to its smooth surface and low friction coefficient.
For daily wig wearers with healthy scalps, these same materials are still preferable. The liner serves a hygiene function, a comfort function, and now, given the emerging science on dermal absorption, a protective function as well.
Avoid Stocking Cap Liners Made from Synthetic Materials
The standard nylon stocking cap liner, which is the most widely available and most commonly used liner in the hair replacement industry, is made from nylon, a synthetic polymer. Wearing a nylon stocking cap between the scalp and a plastic wig base does not reduce chemical exposure. It adds a second plastic layer to the equation.
Nylon stocking caps are also occlusive, trapping heat and moisture against the scalp and creating the warm, hydrated environment that research has linked to higher rates of chemical absorption. They offer no meaningful barrier against the compounds leaching from synthetic cap bases.
Polyester stocking caps carry the same concerns. Any liner made from synthetic fiber, regardless of its softness or texture, should be avoided for clients who wear cranial prosthetics on a daily basis or who have any degree of scalp compromise.
The replacement is simple. Bamboo, organic cotton, or silk cap liners are available, washable, and provide meaningful protection. The switch costs almost nothing and removes a significant variable from the client's daily chemical exposure.
Reduce Wear Duration and Prioritize Scalp Recovery
Duration of contact is a primary driver of chemical absorption across all exposure pathways. The longer a synthetic material sits against the scalp, the greater the potential for transfer. For clients who remove their cranial prosthetic daily, this variable is already controlled. For extended-wear clients, factoring material risk into wear duration recommendations is now a clinical responsibility.
Scalp care during periods without the prosthetic should include gentle cleansing to remove accumulated residue and chemical deposits, followed by application of a barrier-supporting product appropriate to the client's scalp condition.
A Field That Must Evolve
The science on plastic exposure through skin contact is not complete. Researchers are still mapping the mechanisms, dose-response relationships, and long-term health implications. But the direction of the evidence is consistent, and waiting for a definitive verdict before making better decisions for our clients is not a standard we should accept.
The hair replacement industry has a history of adopting materials based on aesthetics and performance without fully evaluating the physiological implications of placing those materials against human skin for extended periods. That approach was adequate when the science was not available to challenge it. The science is now available.
At HIASTI, we believe that every professional serving clients who wear cranial prosthetics has a responsibility to understand what those materials are made of, what the emerging science says about prolonged skin contact, and what practical steps exist to reduce unnecessary exposure. That is not alarmism. That is clinical practice.
The wig is not just a style tool. It is a material that lives against living tissue. We owe it to our clients to treat it accordingly.
©2026 Egypt Lawson, All rights reserved.
No portion of this article or any content on this website-including but not limited to text, images, terminology, research findings, methodologies, or original concepts-may be copied, reproduced, distributed, republished, downloaded, displayed, posted, transmitted, or used in any form or by any means, including photocopying, recording, or other electronic or mechanical methods, without the prior written permission of Hairline Illusions / HIASTI.
This prohibition expressly includes the use of artificial intelligence (Al) tools, large language models, or any automated systems to copy, paraphrase, rewrite, summarize, or create derivative works based on this content.
For permission requests, contact: info@hairlineillusions.com
Unauthorized use may result in legal action.




Comments